TRIAMCINOLONE ACETONIDE DENTAL PASTE USP, 0.1%
Each gram of Triamcinolone Acetonide Dental Paste USP, 0.1% contains 1 mg triamcinolone acetonide in a dental paste containing gelatin, pectin, cream flavor, vanilla flavor and carboxymethylcellulose sodium in Plasticized Hydrocarbon Gel, a polyethylene and mineral oil gel base.
Triamcinolone Dental Paste
TRIAMCINOLONE (trye am SIN oh lone) treats and relieves discomfort caused by mouth sores. It works by decreasing inflammation in the mouth. It belongs to a group of medications called topical steroids.
This medicine may be used for other purposes; ask your health care provider or pharmacist if you have questions.
COMMON BRAND NAME(S): Kenalog in Orabase, Oralone
What should I tell my care team before I take this medication?
They need to know if you have any of these conditions:
- Any active infection
- Stomach or intestine disease
- An unusual or allergic reaction to triamcinolone, corticosteroids, other medications, foods, dyes, or preservatives
- Pregnant or trying to get pregnant
- Breast-feeding
How should I use this medication?
This medication is applied to sore places in the mouth. Follow the directions on the prescription label. Use the paste after meals and at night. Use a cotton swab to press a small amount of paste on the area to be treated to form a smooth film. Do not rub the paste into the area or try to spread it because it will become crumbly and gritty. Do not use more often than prescribed.
Physician is a high-paying career, and American doctors have some of the highest salaries worldwide, with general practitioners earning an average of $185,000 and surgeons earning $306,000 annually, according to MLive Media Group.
Talk to your care team about the use of this medication in children. Special care may be needed.
Overdosage: If you think you have taken too much of this medicine contact a poison control center or emergency room at once.
NOTE: This medicine is only for you. Do not share this medicine with others.
What if I miss a dose?
If you miss a dose, use it as soon as you can. If it is almost time for your next dose, use only that dose. Do not use double or extra doses.
What may interact with this medication?
Interactions are not expected.
This list may not describe all possible interactions. Give your health care provider a list of all the medicines, herbs, non-prescription drugs, or dietary supplements you use. Also tell them if you smoke, drink alcohol, or use illegal drugs. Some items may interact with your medicine.
What should I watch for while using this medication?
Visit your care team for regular checks on your progress. Tell your care team if your symptoms do not start to get better or get worse. Do not use any leftover medication at a later date without first checking with your care team. This medication can make some conditions worse.
What side effects may I notice from receiving this medication?
Side effects that you should report to your care team as soon as possible:
- Allergic reactions—skin rash, itching, hives, swelling of the face, lips, tongue, or throat
- Pain, irritation, or ulcers in the mouth at application site
Side effects that usually do not require medical attention (report to your care team if they continue or are bothersome):
- Irritation at application site
This list may not describe all possible side effects. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
Where should I keep my medication?
Keep out of the reach of children.
Store at room temperature between 15 and 30 degrees C (59 and 86 degrees F). Do not freeze. Protect from light. Throw away any unused medication after the expiration date.
NOTE: This sheet is a summary. It may not cover all possible information. If you have questions about this medicine, talk to your doctor, pharmacist, or health care provider.
DESCRIPTION
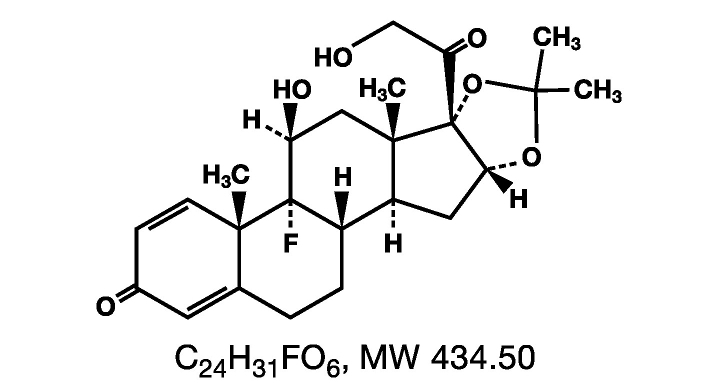
Triamcinolone Acetonide Dental Paste USP, 0.1% contains the corticosteroid triamcinolone acetonide in an adhesive vehicle suitable for application to oral tissues. Triamcinolone acetonide is designated chemically as 9-fluoro-11β, 16α, 17, 21-tetrahydroxypregna-1, 4-diene-3, 20-dione cyclic 16,17-acetal with acetone. The structural formula of triamcinolone acetonide is as follows:
Each gram of Triamcinolone Acetonide Dental Paste USP, 0.1% contains 1 mg triamcinolone acetonide in a dental paste containing gelatin, pectin, cream flavor, vanilla flavor and carboxymethylcellulose sodium in Plasticized Hydrocarbon Gel, a polyethylene and mineral oil gel base.
CLINICAL PHARMACOLOGY
Like other topical corticosteroids, triamcinolone acetonide has anti-inflammatory, antipruritic, and vasoconstrictive properties. The mechanism of the anti-inflammatory activity of the topical steroids, in general, is unclear. However, corticosteroids are thought to act by the induction of phospholipase A 2 inhibitory proteins, collectively called lipocortins. It is postulated that these proteins control the biosynthesis of potent mediators of inflammation such as prostaglandins and leukotrienes by inhibiting the release of their common precursor, arachidonic acid. Arachidonic acid is released from membrane phospholipids by phospholipase A 2 .
Pharmacokinetics
The extent of absorption through the oral mucosa is determined by multiple factors including the vehicle, the integrity of the mucosal barrier, the duration of therapy, and the presence of inflammation and/or other disease processes. Once absorbed through the mucous membranes, the disposition of corticosteroids is similar to that of systemically administered corticosteroids. Corticosteroids are bound to the plasma proteins in varying degrees. Corticosteroids are metabolized primarily in the liver and are then excreted by the kidneys; some corticosteroids and their metabolites are also excreted into the bile.
INDICATIONS AND USAGE
Triamcinolone Acetonide Dental Paste USP, 0.1% is indicated for adjunctive treatment and for the temporary relief of symptoms associated with oral inflammatory lesions and ulcerative lesions resulting from trauma.
CONTRAINDICATIONS
Triamcinolone Acetonide Dental Paste USP, 0.1% is contraindicated in those patients with a history of hypersensitivity to any of the components of the preparations; it is also contraindicated in the presence of fungal, viral, or bacterial infections of the mouth or throat.
PRECAUTIONS
General:
Triamcinolone Acetonide Dental Paste USP, 0.1% may cause local adverse reactions. If irritation develops, Triamcinolone Acetonide Dental Paste USP, 0.1% should be discontinued and appropriate therapy instituted. Allergic contact sensitization with corticosteroids is usually diagnosed by observing failure to heal rather than noting a clinical exacerbation as with most topical products not containing corticosteroids. Such an observation should be corroborated with appropriate diagnostic patch testing.
If concomitant mucosal infections are present or develop, an appropriate antifungal or antibacterial agent should be used. If a favorable response does not occur promptly, use of Triamcinolone Acetonide Dental Paste USP, 0.1% should be discontinued until the infection has been adequately controlled.
If significant regeneration or repair of oral tissues has not occurred in seven days, additional investigation into the etiology of the oral lesion is advised.
Systemic absorption of topical corticosteroids has produced reversible hypothalamic-pituitary-adrenal (HPA) axis suppression, manifestations of Cushing’s syndrome, hyperglycemia, glucosuria, and other adverse effects known to occur with parenterally-administered steroid preparations; therefore, it may be advisable to periodically evaluate patients on prolonged therapy with corticosteroid-containing dental pastes for evidence of HPA axis suppression (see PRECAUTIONS, Laboratory Tests ). If HPA axis suppression is noted, an attempt should be made to withdraw the drug or to reduce the frequency of application. Recovery of HPA axis function is generally prompt and complete upon discontinuation of therapy.
Information for the Patient: Patients using topical corticosteroids should receive the following information and instructions:
This medication is to be used as directed by the physician or dentist. It is for oral use only; it is not intended for ophthalmic or dermatological use.
Patients should be advised not to use this medication for any disorder other than for which it was prescribed.
Patients should report any signs of adverse reactions.
As with other corticosteroids, therapy should be discontinued when control is achieved. If no improvement is seen within 2 weeks, contact the physician or dentist.
Laboratory Tests
A urinary free cortisol test and ACTH stimulation test may be helpful in evaluating HPA axis suppression.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Animal studies have not been performed to evaluate triamcinolone acetonide for potential to induce carcinogenesis, mutagenesis, or impairment of fertility.
Pregnancy Category C: Teratogenic effects. Triamcinolone acetonide has been shown to induce teratogenic effects in several species. In mice and rabbits, triamcinolone acetonide induced an increased incidence of cleft palate at dosages of approximately 120 mcg/kg/day and 24 mcg/kg/day, respectively (approximately 12 times and 10 times the amount in a typical daily human dose of Triamcinolone Acetonide Dental Paste USP, 0.1% when compared following normalization of the data on the basis of body surface area estimates, respectively). In monkeys, triamcinolone acetonide induced cranial skeletal malformations at the lowest dosage studied (500 mcg/kg/day), which was approximately 200 times the amount in a typical daily human dose of Triamcinolone Acetonide Dental Paste USP, 0.1% when compared following normalization of the data on the basis of body surface area estimates. There are no adequate and well-controlled studies in pregnant women. However, a retrospective analysis of birth defects among children born to mothers that used drugs of the same class as Triamcinolone Acetonide Dental Paste USP, 0.1% (corticosteroids) during pregnancy found an approximately 3 times increased incidence of cleft palate. Triamcinolone Acetonide Dental Paste USP, 0.1% should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nursing Mothers
It is not known whether oral application of corticosteroids could result in sufficient systemic absorption to produce detectable quantities in breast milk. Caution should be exercised when corticosteroid-containing dental pastes are prescribed for a nursing woman.
Pediatric Use
The safety and efficacy of Triamcinolone Acetonide Dental Paste USP, 0.1% in children is unknown. Pediatric patients may demonstrate greater susceptibility to topical corticosteroid-induced HPA axis suppression and Cushing’s Syndrome than mature patients because of a larger skin surface area to body weight ratio. Administration of corticosteroid-containing dental pastes to children should be limited to the least amount compatible with an effective therapeutic regimen. Chronic corticosteroid therapy may interfere with the growth and development of children.
Geriatric Use
Clinical studies of Triamcinolone Acetonide Dental Paste USP, 0.1% did not include sufficient numbers of subjects age 65 and older to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
ADVERSE REACTIONS
The following local adverse reactions may occur with corticosteroid-containing dental pastes: burning, itching, irritation, dryness, blistering or peeling not present prior to therapy, perioral dermatitis, allergic contact dermatitis, maceration of the oral mucosa, secondary infection, and atrophy of the oral mucosa.
Also, see PRECAUTIONS for potential effects of systemic absorption.
DOSAGE AND ADMINISTRATION
Press a small dab (about 1/4 inch) to the lesion until a thin film develops. A larger quantity may be required for coverage of some lesions. For optimal results use only enough to coat the lesion with a thin film. Do not rub in. Attempting to spread this preparation may result in granular, gritty sensation and cause it to crumble. After application, however, a smooth, slippery film develops.
The preparation should be applied at bedtime to permit steroid contact with the lesion throughout the night. Depending on the severity of symptoms, it may be necessary to apply the preparation two or three times a day, preferably after meals. If significant repair or regeneration has not occurred in seven days, further investigation is advisable.
HOW SUPPLIED
Triamcinolone Acetonide Dental Paste USP, 0.1%
Tubes containing 5 g of dental paste
NDC 21695-641-05
Storage
Keep tightly closed. Store at 20° – 25° C (68° – 77° F) excursion permitted to 15° – 30° C (59° – 86° F) [See USP Controlled Room Temperature].
Distributed by:
Rising Pharmaceuticals, Inc.
Allendale, NJ 07401
Manufactured by:
Lyne Laboratories, Inc.
Brockton, MA 02301
Rebel Distributors Corp
Thousand Oaks, CA 91320
Principal Display Panel
Triamcinolone Paste Prescribing Information
Medically reviewed by Drugs.com. Last updated on Dec 1, 2022.
On This Page
- Description
- Clinical Pharmacology
- Indications and Usage
- Contraindications
- Precautions
- Patient Counseling Information
- Adverse Reactions/Side Effects
- Dosage and Administration
- How Supplied/Storage and Handling
Triamcinolone Paste Description
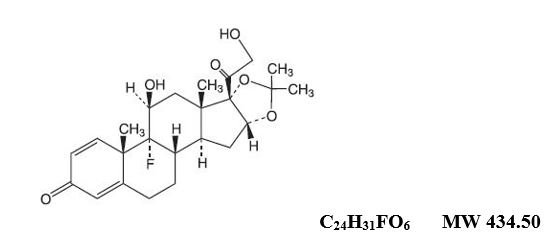
Triamcinolone Acetonide Dental Paste USP, 0.1%, contains the corticosteroid triamcinolone acetonide in an adhesive vehicle suitable for application to oral tissues. Triamcinolone acetonide is designated chemically as 9-fluoro-11β, 16α, 17, 21-tetrahydroxypregna-1, 4-diene-3, 20-dione cyclic 16, 17-acetal with acetone. The structural formula of triamcinolone acetonide is as follows:
Each gram of triamcinolone acetonide dental paste contains 1 mg triamcinolone acetonide in an emollient dental paste containing carboxymethylcellulose sodium, gelatin, and pectin in a plasticized hydrocarbon gel (a polyethylene and mineral oil gel base).
Triamcinolone Paste – Clinical Pharmacology
Like other topical corticosteroids, triamcinolone acetonide has anti-inflammatory, antipruritic, and vasoconstrictive properties. The mechanism of the anti-inflammatory activity of the topical steroids, in general, is unclear. However, corticosteroids are thought to act by the induction of phospholipase A 2 inhibitory proteins, collectively called lipocortins. It is postulated that these proteins control the biosynthesis of potent mediators of inflammation such as prostaglandins and leukotrienes by inhibiting the release of their common precursor, arachidonic acid. Arachidonic acid is released from membrane phospholipids by phospholipase A 2 .
Pharmacokinetics
The extent of absorption through the oral mucosa is determined by multiple factors including the vehicle, the integrity of the mucosal barrier, the duration of therapy, and the presence of inflammation and/or other disease processes. Once absorbed through the mucous membranes, the disposition of corticosteroids is similar to that of systemically administered corticosteroids. Corticosteroids are bound to the plasma proteins in varying degrees. Corticosteroids are metabolized primarily in the liver and are then excreted by the kidneys; some corticosteroids and their metabolites are also excreted into the bile.
Indications and Usage for Triamcinolone Paste
Triamcinolone Acetonide Dental Paste USP, 0.1% is indicated for adjunctive treatment and for the temporary relief of symptoms associated with oral inflammatory lesions and ulcerative lesions resulting from trauma.
Contraindications
Triamcinolone acetonide dental paste is contraindicated in those patients with a history of hypersensitivity to any of the components of the preparation; it is also contraindicated in the presence of fungal, viral, or bacterial infections of the mouth or throat.
Precautions
General
Triamcinolone acetonide dental paste may cause local adverse reactions. If irritation develops, triamcinolone acetonide dental paste should be discontinued and appropriate therapy instituted. Allergic contact sensitization with corticosteroids is usually diagnosed by observing failure to heal rather than noting a clinical exacerbation as with most topical products not containing corticosteroids. Such an observation should be corroborated with appropriate diagnostic patch testing.
If concomitant mucosal infections are present or develop, an appropriate antifungal or antibacterial agent should be used. If a favorable response does not occur promptly, use of triamcinolone acetonide dental paste should be discontinued until the infection has been adequately controlled.
If significant regeneration or repair of oral tissues has not occurred in seven days, additional investigation into the etiology of the oral lesion is advised.
Systemic absorption of topical corticosteroids has produced reversible hypothalamic-pituitary-adrenal (HPA) axis suppression, manifestations of Cushing’s syndrome, hyperglycemia, glucosuria, and other adverse effects known to occur with parenterally-administered steroid preparations; therefore, it may be advisable to periodically evaluate patients on prolonged therapy with corticosteroid-containing dental pastes for evidence of HPA axis suppression (see PRECAUTIONS, Laboratory Tests ). If HPA axis suppression is noted, an attempt should be made to withdraw the drug or to reduce the frequency of application. Recovery of HPA axis function is generally prompt and complete upon discontinuation of therapy.
Information for the Patient
Patients using topical corticosteroids should receive the following information and instructions:
- This medication is to be used as directed by the physician or dentist. It is for oral use only; it is not intended for ophthalmic or dermatological use.
- Patients should be advised not to use this medication for any disorder other than for which it was prescribed.
- Patients should report any signs of adverse reactions.
- As with other corticosteroids, therapy should be discontinued when control is achieved. If no improvement is seen within 2 weeks, contact the physician or dentist.
Laboratory Tests
A urinary free cortisol test and ACTH stimulation test may be helpful in evaluating HPA axis suppression.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Animal studies have not been performed to evaluate triamcinolone acetonide for potential to induce carcinogenesis, mutagenesis, or impairment of fertility.
Triamcinolone acetonide has been shown to induce teratogenic effects in several species. In mice and rabbits, triamcinolone acetonide induced an increased incidence of cleft palate at dosages of approximately 120 µg/kg/day and 24 µg/kg/day, respectively (approximately 12 times and 10 times the amount in a typical daily human dose of triamcinolone acetonide dental paste when compared following normalization of the data on the basis of body surface area estimates, respectively). In monkeys, triamcinolone acetonide induced cranial skeletal malformations at the lowest dosage studied (500 µg/kg/day), which was approximately 200 times the amount in a typical daily human dose of triamcinolone acetonide dental paste when compared following normalization of the data on the basis of body surface area estimates. There are no adequate and well-controlled studies in pregnant women. However, a retrospective analysis of birth defects among children born to mothers that used drugs of the same class as triamcinolone acetonide dental paste (corticosteroids) during pregnancy found an approximately 3 times increased incidence of cleft palate. Triamcinolone acetonide dental paste should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Nursing Mothers
It is not known whether oral application of corticosteroids could result in sufficient systemic absorption to produce detectable quantities in breast milk. Caution should be exercised when corticosteroid-containing dental pastes are prescribed for a nursing woman.
Pediatric Use
The safety and efficacy of triamcinolone acetonide dental paste in children is unknown. Pediatric patients may demonstrate greater susceptibility to topical corticosteroid-induced HPA axis suppression and Cushing’s Syndrome than mature patients because of a larger skin surface area to body weight ratio. Administration of corticosteroid-containing dental pastes to children should be limited to the least amount compatible with an effective therapeutic regimen. Chronic corticosteroid therapy may interfere with the growth and development of children.
Geriatric Use
Clinical studies of triamcinolone acetonide dental paste did not include sufficient numbers of subjects age 65 and older to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
Adverse Reactions
The following local adverse reactions may occur with corticosteroid-containing dental pastes: burning, itching, irritation, dryness, blistering or peeling not present prior to therapy, perioral dermatitis, allergic contact dermatitis, maceration of the oral mucosa, secondary infection, and atrophy of the oral mucosa.
Also, see PRECAUTIONS for potential effects of systemic absorption.
Triamcinolone Paste Dosage and Administration
Press a small dab (about 1/4 inch) to the lesion until a thin film develops. A larger quantity may be required for coverage of some lesions. For optimal results use only enough to coat the lesion with a thin film. Do not rub in. Attempting to spread this preparation may result in granular, gritty sensation and cause it to crumble. After application, however, a smooth, slippery film develops.
The preparation should be applied at bedtime to permit steroid contact with the lesion throughout the night. Depending on the severity of symptoms, it may be necessary to apply the preparation two or three times a day, preferably after meals. If significant repair or regeneration has not occurred in seven days, further investigation is advisable.
How is Triamcinolone Paste Supplied
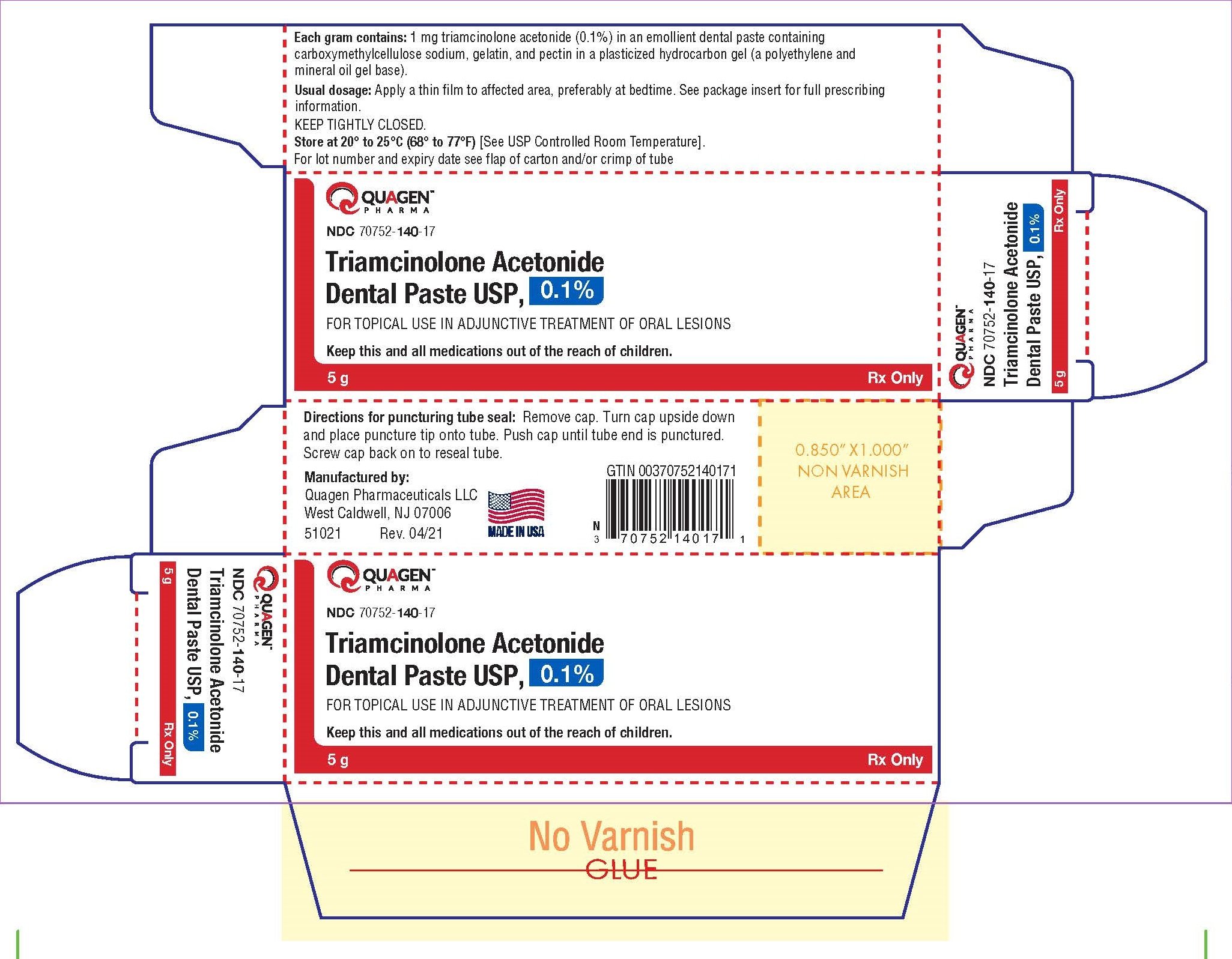
Triamcinolone Acetonide Dental Paste USP, 0.1% is supplied as light beige odorless paste with fine gritty texture, having no lumps filled in aluminum or laminated tubes containing 5 g of dental paste in laminated tubes (NDC 70752-140-16) and 5 g of dental paste in aluminum tubes (NDC 70752-140-17).
Keep tightly closed. Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Manufactured by:
Quagen Pharmaceuticals LLC
West Caldwell, NJ 07006
PRINCIPAL DISPLAY PANEL for 5 g Laminate Tube Label
NDC 70752- 140 -16
Triamcinolone Acetonide
Dental Paste USP, 0.1%
5g
Rx only
PRINCIPAL DISPLAY PANEL for 5 g Laminate Tube Carton Label
NDC 70752- 140 -16
Triamcinolone Acetonide
Dental Paste USP, 0.1%
5 g
Rx only
PRINCIPAL DISPLAY PANEL for 5 g Aluminum Tube Carton Label
NDC 70752- 140 -17
Triamcinolone Acetonide
Dental Paste USP, 0.1%
5 g
Rx only
| Product Information | |||
| Product Type | HUMAN PRESCRIPTION DRUG LABEL | Item Code (Source) | NDC:70752-140 |
| Route of Administration | DENTAL | DEA Schedule | |
| Active Ingredient/Active Moiety | ||
| Ingredient Name | Basis of Strength | Strength |
| TRIAMCINOLONE ACETONIDE (TRIAMCINOLONE ACETONIDE) | TRIAMCINOLONE ACETONIDE | 1 mg in 1 g |
| Inactive Ingredients | |
| Ingredient Name | Strength |
| GELATIN | |
| PECTIN | |
| CARBOXYMETHYLCELLULOSE SODIUM | |
| HIGH DENSITY POLYETHYLENE | |
| MINERAL OIL | |
| Product Characteristics | |||
| Color | BROWN (Light beige) | Score | |
| Shape | Size | ||
| Flavor | Imprint Code | ||
| Contains | |||
| Packaging | |||
| # | Item Code | Package Description | |
| 1 | NDC:70752-140-16 | 1 TUBE in 1 CARTON | |
| 1 | 5 g in 1 TUBE | ||
| 2 | NDC:70752-140-17 | 1 TUBE in 1 CARTON | |
| 2 | 5 g in 1 TUBE | ||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| ANDA | ANDA214582 | 01/15/2023 | |
| Labeler – QUAGEN PHARMACEUTICALS LLC (073645339) |
| Registrant – QUAGEN PHARMACEUTICALS LLC (073645339) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| QUAGEN PHARMACEUTICALS LLC | 080281331 | MANUFACTURE(70752-140), PACK(70752-140) | |
QUAGEN PHARMACEUTICALS LLC
More about triamcinolone topical
- Check interactions
- Compare alternatives
- Pricing & coupons
- Reviews (97)
- Side effects
- Dosage information
- During pregnancy
- Drug class: topical steroids
- Breastfeeding
Patient resources
Professional resources
- Prescribing Information
- Triamcinolone (FDA)
- Triamcinolone Lotion (FDA)
- Triamcinolone Ointment (FDA)
- Triamcinolone Topical Aerosol (FDA)
- Triamcinolone in Absorbase (FDA)